Research
Shining light on sustainable organic synthesis and illuminating the origins of life.
Green Organic Solvents for Conventional Synthesis
In conventional organic synthesis, some of the most frequently performed reaction classes require the use of hazardous organic solvents like dichloromethane (DCM) and N,N-dimethylformamide (DMF). We identify and employ new organic solvents that are biodegradable and safer than these traditional solvents. These new green solvents often outperform the original solvent in reactions of industrial interest. Of particular interest are amide coupling reactions, the most frequently performed reaction class in medicinal organic synthesis.

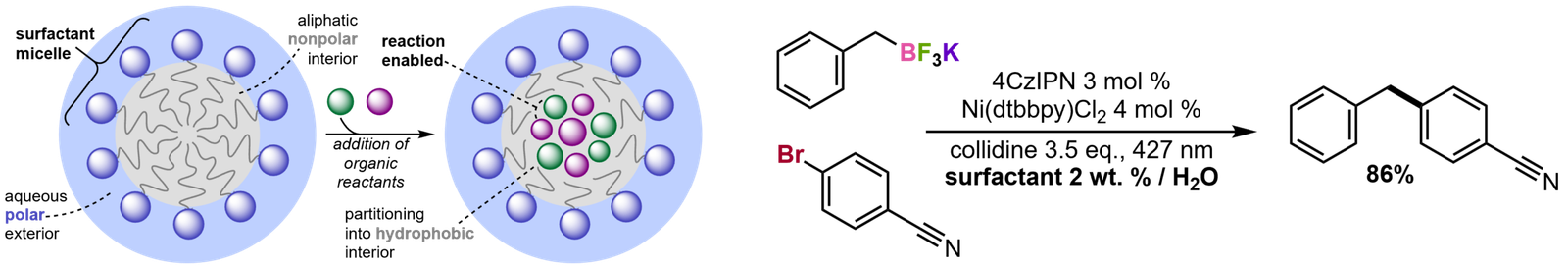
Surfactants for Photoredox Catalysis in Water
Photoredox catalysis is a subfield of organic synthesis that uses light to enable chemical transformations which would otherwise be difficult or impossible by traditional synthetic methods. Photoredox reactions are typically performed in hazardous organic solvents like DMF. To obviate these organic solvents and switch to more sustainable methods of synthesis, we have developed new biodegradable, redox-inactive surfactants that enable photoredox cross-coupling reactions in water. The surfactants accomplish this by emulsifying the water-insoluble organic compounds, allowing them to diffuse and react.

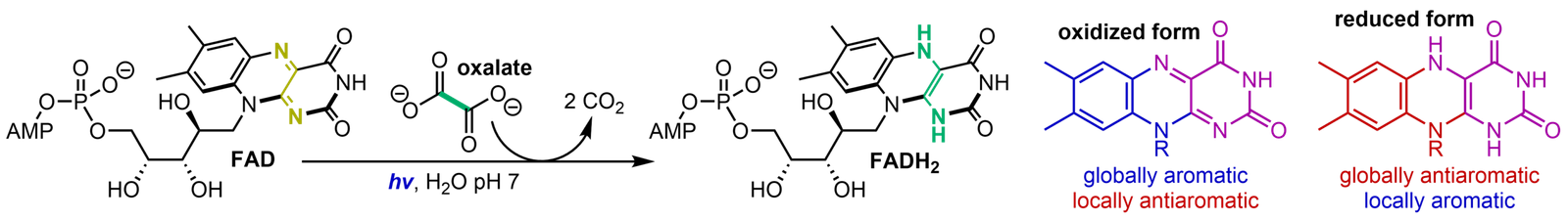
Origins of Life
How life began on the early Earth is one of the greatest unsolved problems in science. A central question in the study of the origin of life is the emergence of the nucleic acids, DNA and RNA, and their constituent nucleotides. Related to this is the chemical origins of the redox-active dinucleotides NAD and FAD. The redox-active “nucleobases” of these nucleotides have precise structures that determine their reactivity. Flavin, the redox-active component of FAD, has a complex tricyclic structure that is highly photoreactive. We are interested in the chemical etiology of this structure and its possible coevolution with nicotinamide nucleotides and the canonical RNA nucleotides.